
Se acerca el momento para este precio al que he dedicado tantas y tantas lineas. Exactamente a un mes vista, el próximo día 14 de abril, sabremos si la FDA, da el OK definitivo o no, para Afrezza; Recordemos, un polvo para inhalación que servirá para mantener a raya el control glucémico en pacientes adultos que sufren de diabetes tipo I o de tipo II.
Quienes desee embeberse mas sobre todo ello, puede acudir a su PÁGINA WEB, y ponerse al día tanto sobre Afrezza, como del resto de su «pipeline«, que por cierto, es bastante interesante.
Os resumo de manera gráfica:
Diabetes
Afrezza se encuentra en fase avanzada de investigación clínica para el tratamiento de adultos con diabetes tipo 1 o diabetes tipo 2 para el control de la hiperglucemia. Para obtener más información, visitar www.clinicaltrials.gov .
Cáncer
MKC1106 – MT está siendo evaluado en un ensayo clínico de fase 2 en pacientes con melanoma avanzado.
Para obtener más información, visite www.clinicaltrials.gov
Manuel, nuestro doctor particular, nos relataba las bondades y espléndido futuro que este valor podía tener, sobre todo en lo relativo a su medicamento “Afrezza” para una forma diferente de tratar la diabetes.
En sus palabras:
«Recordemos que en la actualidad la forma de administrarse la insulina por parte de todos los pacientes de diabetes tipo 1 y 2, es a través de inyección.
Con Afrezza, el cambio sería revolucionario ya que consistiría en un inhalador dosificador, con la dosis programada y con un cartucho para un solo uso.
Parece que el consenso casi unánime de los expertos del sector, predice que este fármaco, será aprobado en 2014 y le otorgan o predestinan un mercado potencial de mas de 3 mil millones de dólares anuales. Hecho que pone en la mirilla a Mannkin Corporation, de algunas grandes farmacéuticas”.
Una vez situados, debemos recordar que el desarrollo de AFREZZA, le ha costado a MANNKIND, la bonita cifra de 2.400 millones de dólares. Imaginar lo costoso de todo esto y lo rápido que se podría amortizar con ese mercado potencial.
Bien, ahora vamos con lo que ocurriría en caso de la FDA, decidiera dar el «SI» para Afrezza, teniendo en cuenta las apuestas con esta cotización, esto es los apostantes a corto y los situados largos.
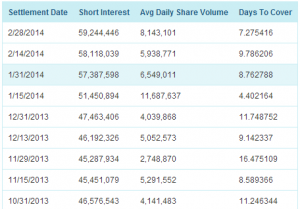
Para ello os dejo la gráfica lateral adjunta, según la cual, a 28 de febrero, osea, hace nada, tendríamos 59.244.446 acciones, «vendidas«.
Sobre 377 millones de acciones en circulación de la compañía, calculen ustedes el «cortocircuito» que se produciría, caso de aprobación para Afrezza. Lo que correrían los cortos para tratar de liquidar sus posiciones y donde llevaría ese ímpetu a la cotización. Claro que si la decisión fuera la contraria, prepárense para recibir la del pulpo, aquellos comprados en el precio.
Contemplando sus gráficos, yo diría, DE SER UN PRECIO NORMAL, que tiene toda la pinta de querer, pero no pertenecen sus gráficos a un valor que pudiéramos llamar «NORMAL«. Ya saben ustedes como se las gastan, como juegan, como amagan hacia un lado, para después dirigirse al contrario. ¿Qué les voy a contar a demás sobre toda las news no precisamente buenas, sobre los miembros de la FDA?. ¿Qué les diría sobre las guerras larvadas entre las multinacionales farmacéuticas?. ¿Qué les podría añadir, sobre los intereses previamente pagados, de los articulistas o escribientes en los blogs americanos?.
Creo como siempre, cuando se trata de tomar partido en cualquiera de estos precios, debemos poner al margen sus gráficos, su aspecto técnico y centrarnos en el producto.
Si hacemos eso, si ustedes se informan cuanto me he informado yo, en lo negativo y en lo positivo, podrán tomar una decisión:
Decidirán si compran o no, acciones de esta empresa para su cartera.
Me pronunciaré, como siempre, aún a riesgo de equivocarme, como en muchas ocasiones, por fortuna las menos, sobre este precio. PARA MI ES COMPRA. No se si llegaré a tiempo para picar algo, pero todo se andará por el camino.
Suerte con ese dedo y estén muy atentos a este precio en las próximas semanas. ¡Su futuro, está muy cerca!.
……………………………………………………………………………………………………….
………………………………………………………………………………………………………
ACTUALIZADO 22 DE MARZO DE 2014.
…………………………………………………………………………………………………….
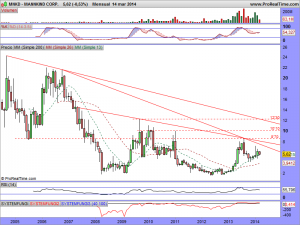
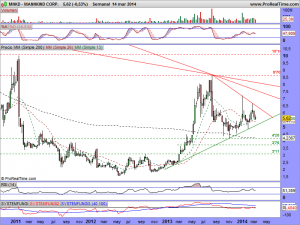
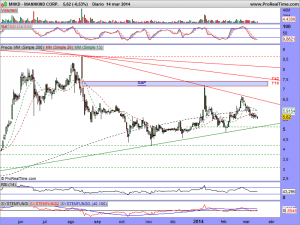
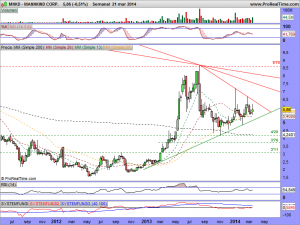
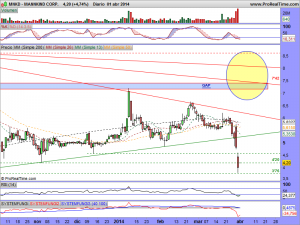
Les actualizo su gráfico en semanal, donde se puede observar las ganas que tiene de romper la cotización, al estar sometida dentro de un rango cada vez menor….
………………………………………………………………………………………….
ACTUALIZADO 27 DE MARZO DE 2014.
…………………………………………………………………………………………………………………………..
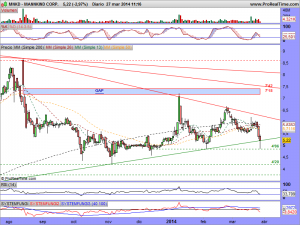
Estamos a poco mas de quince días del veredicto, y van comenzando a calentarse motores……….
…………………………………………………………………………………………………….
ACTUALIZADO A 31 DE MARZO DE 2014.
…………………………………………………………………………………………………………
¡Que difícil es ganar dinero con una «pharma«!. Ya ven ustedes el sopapo que lleva en el cuerpo un día si y otro también en espera de la decisión del panel de expertos. No he leído que se hayan pronunciado, pero la manipulación es tremenda, como ven ustedes…………..
Les deseo toda la suerte del mundo a todos los que estén comprados en este «tobogan».
Así está el gráfico a estas horas…………
…………………………………………………………………………………………….
ACTUALIZADO 02 DE ABRIL DE 2014.
………………………………………………………………………………………………..
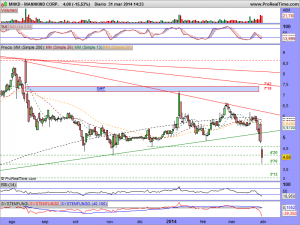
¡Enhorabuena a los afortunados!, el comité de expertos ha dado su ok, provisional para la posterior aprobación del producto.
Una vez mas, soy feliz por haber decidido en su día, colgar este precio, que tanto dinero les hará ganar a todos aquellos que decidieron comprar.
Disfruten del momento y ¡que continúe la cabalgada!…..
………………………………………………………………………………………………………
Les vuelvo a actualizar su gráfico, ahora con el mercado abierto, ¡ah! y presten atención a lo milimétricos que han sido de momento con esa bajista y entenderán el porqué estaba precisamente ahí colocada……..
………………………………………………………………………………………………………………..




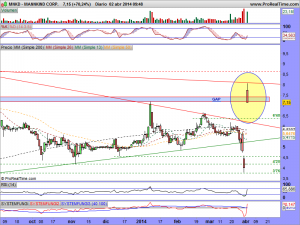
MNKD-Espero y deseo por el avance que supone para los pacientes diabèticos, que salga adelante.
Para los inversores del foro, les digo que no es la primera vez que se intenta comercializar un fàrmaco de administración inhalada para controlar la diabetes.
Ya Pfizer hace 7 años comercializó Exubera, que despuès de estar aprobado y comercializado ,fuè un fracaso , que acabó retirando del mercado.Aparte de los problemas de seguridad que aparecieron, desde un punto de vista comercial, no fuè facil su introducciòn , puesto que no eliminaba completamente los pinchazo se insulina.
Otras grandes farmacèuticas como Lilly y Novo Nordisk, tambièn eliminaron sus desarrollos en este campo.
Aunque es de hace unos años, os adjunto el enlace a la noticia.
http://www.cnnexpansion.com//negocios/2008/04/09/pfizer-halla-cancer-con-insulina-exubera
Yo estoy metido de lleno, pero no tengo mucha confianza ….
MNKD
Como siempre, quien quiera peces a mojarse.
Gracias SUPER por los comentarios.
O todo o nada con afrezza..y no pinta bien … (
(
http://m.gurufocus.com/news_read.php?id=251485
MNKD & Mark
No entiendo bien el artículo ¿por que no pinta bien?
Mil gracias.
MNKD-Hola Jose , te traduzco una pequeña parte del articulo…
«En el pasado, otras insulinas inhalados no han sido muy exitosos. En 2006, Pfizer (PFE) lanza Exubera, el primera aprobados por la FDA inhalado insulina. Pfizer dejó de comercialización de la droga en octubre de 2007 como sólo había hecho $12 millones en ventas en los tres primeros trimestres del año. Otras compañías también decidieron suspender sus propios programas de insulina inhalada. Esto significa que incluso si Afrezza despeja su camino a la aprobación de la FDA y lo convierte en el mercado, será difícil convencer a los médicos a prescribir este medicamento sobre otros existentes y «más seguros». Además, tendrá que competir con los líderes del mercado, tales como Novo Nordisk (NVO) que se deriva el 78 por ciento de las ventas totales de productos de cuidado de la diabetes.»
Como ves, incluso aunque se apruebe, las expectativas , no son muy buenas.Saca tus propias conclusiones.
MNKD
Gracias JULI, algo de eso entendi.
SA AnalysisMLV Sees A Good Chance Of FDA Approval For MannKind’s Afrezza And 60% Upsidemar 18 2014, 11:53 | by Ben Yoffe about: MNKDSummaryIn this interview, MLV’s Dr. Graig Suvannavejh discusses his views on the future of MannKind as it gets closer to the upcoming AdCom and PDUFA for Afrezza.Dr. Suvannavejh believes Afrezza has 90% chance of a positive FDA AdCom outcome.Additionally, we discuss current investor concerns, including; the regulatory outlook and commercial concerns.MannKind (MNKD) faces an FDA Endocrinologic and Metabolic Drugs Advisory Committee meeting on April 1, 2014, for the review of the company’s New Drug Application (NDA) for Afrezza, an ultra rapid-acting insulin developed for the treatment of adult patients with type 1 or type 2 diabetes. Obviously, if the committee will recommend the drug for approval, it could impact the stock price in a significant manner. However, there was a lot of skepticism around the company and the stock in the past few years. MannKind has twice received Complete Response letters (CRLs) from the FDA when seeking approval for Afrezza. Following discussions with the FDA, the company has embarked on two Phase 3 trials that leverage their new dry powder that is considerably simpler for diabetics to use. On August 14, 2013, the company announced that both Phase 3 trials met their primary endpoints, along with the majority of their secondary endpoints. So now all eyes will be turning to the committee votes and to the final decision.Last week, I had the pleasure of interviewing the head of healthcare research at MLV & Co. and a highly respected Senior Analyst, Dr. Graig Suvannavejh, which has been covering MannKind since last year. Dr. Suvannavejh has over 10 years of experience as a sell-side analyst covering the biotechnology, specialty pharmaceuticals, and large cap pharmaceuticals sectors. Prior to joining MLV, he was a senior analyst covering specialty pharmaceuticals at Jefferies, and before that, a senior analyst covering biotechnology at UBS. He comes from a strong academic background, holding a Ph.D. in Neuroscience from Northwestern University, and an M.S. in Physiology and a B.S. in Biology, both from Georgetown University.MLV has provided the company with investment banking services in the past, with an ATM agreement that was put in place in March 2013, and another in March 2014.Ben Yoffe: Dr. Suvannavejh, the next most significant catalysts for MannKind are the April 1 Advisory Committee meeting for Afrezza, followed by the April 15 Prescription Drug User Fee Act (PDUFA) date. What are your expectations?Dr. Suvannavejh: The April 1 AdCom was a surprise to many of us, including myself. But I really see this as more of a «checking the box» exercise by FDA, especially given a recent change in the leadership at the FDA’s Endocrinology and Metabolic Drugs division. Also, there have been some safety issues that have been associated with a number of high-profile diabetes drugs over the past several years. Keep in mind that diabetics are on their meds in essence for the rest of their lives, and while efficacy is certainly important, safety is more so. So perhaps the new head of the division is being a bit conservative for the sake of simply being conservative. After all, there’s no real downside to holding an AdCom from an FDA perspective.In the case of Afrezza, I am not particularly concerned about the outcome of the AdCom given my view that there is nothing worrisome about either the Phase III data itself for Afrezza or the safety profile of the product. A key concern for FDA when it comes to diabetes drugs in the recent past has been the potential increase in cardiovascular adverse events, but from what we’ve seen from the safety profile of Afrezza, there’s no obvious signal. Lastly, what also gives me confidence in a positive AdCom outcome is that Exubera, the very first inhaled insulin product that was approved in 2006, went before an AdCom in September 2005, and it garnered 7-2 votes in favor of FDA approval in both Type I and Type II patients. So unless I’m missing something major, I’ll say there is a 90% chance of a positive FDA AdCom outcome. The caveat is that the FDA’s briefing documents for the AdCom come out on March 28, two business days before the meeting is scheduled to take place. So I’ll have a much better sense of a potential positive outcome then.As for the April 15 PDUFA, there are two issues that I see: first, the issue of an ultimate FDA thumbs up or thumbs down; and second, the issue of whether the April 15 PDUFA gets delayed or not given the close proximity of the April 1 AdCom. As far as I know, at this point, FDA has not informed MNKD whether it will delay its April 15 approval decision deadline. That said, in my opinion, if they chose to do so, I wouldn’t see this as a very big deal, especially if you keep in perspective that the FDA had MNKD conduct an all new and full Phase III program back in 2011, several years after positive data from the initial Phase III trials were already in hand. The issue of two prior complete response letters resulted in essentially a four year delay. So what’s another few months or so? But as for my expectations for ultimate FDA approval, I would peg the odds of success at about 80-90%, fully acknowledging that when it comes to the FDA and decisions it makes, particularly on drug approvals and what may be happening behind the scenes, anything goes.Yoffe: What are the key investor concerns regarding Afrezza’s regulatory and commercial outlook, and what is your opinion on these issues?Dr. Suvannavejh: As far as the regulatory outlook, I think investors have a few concerns. First, there’s a bear thesis out there that the data generated from the most recent Phase III trials for Afrezza don’t adequately address FDA’s concerns that arose from the second complete response letter that was issued in January 2010. My view on this is that while an interesting take, I derive my confidence in the fact that the latest Phase III trials for Afrezza – AFFINITY-1 and AFFINITY-2 – were designed specifically with extensive input from the FDA, and since the data in totality were very positive, at least in my view, I don’t think there will be an issue.A second more theoretical regulatory concern relates to a view that the FDA simply may not want to approve another inhaled insulin product, based on the unknown long-term safety consequences with an insulin product that will be inhaled daily by a diabetic for the rest of his/her life – especially when one keeps in mind that insulin is a known growth factor. My response to that is that every drug that the FDA approves theoretically has unknown long-term safety consequences. This is the exact reason why FDA requires companies to conduct long-term surveillance on newly approved therapies. And so it’s my full expectation that assuming Afrezza is approved, MNKD and/or its potential commercialization partner will absolutely be required to carry out some sort of long-term study as a way to stay on top of any potential safety signals with Afrezza that may arise.In my opinion, it’s really the commercial outlook that’s likely the bigger investor concern. There are two aspects. First, the prior inhaled insulin, Exubera, turned out to be a major commercial failure – Pfizer (PFE) ended up withdrawing that product from the market due to lack of sales and ended up taking a $2 billion write-down on it – and so there is the concern that Afrezza might be a flop as well. My take is that there are many good reasons why Exubera didn’t succeed and many good reasons why Afrezza shouldn’t follow in Exubera’s footsteps.First, the inhaler device for Exubera was simply huge. Anyone who’s familiar with the Exubera device knows that was big, bulky, unattractive, and not discrete in any way; it had a lot of moving parts, and it also required multiple steps for washing and cleaning. So overall, it just wasn’t very patient-friendly. Also, in an attempt to recoup the investment that went into developing the product, Pfizer priced Exubera at a significant premium, reportedly as high as $5 a day compared to the current $2-$3 per day for injected insulin. Exubera was also dosed in terms of grams (when insulin is more typically dosed in units). Lastly, by the time Exubera was finally available commercially, the product was linked to a potential increase in cancer. So not good. To be fair, a causal relationship however was never formally established.I’m not expecting Afrezza to face the exact same problems. First of all, the current Afrezza inhaler, known as Dreamboat, is small and discrete enough to fit in the palm of one’s hand. Also, MNKD has said that the price of Afrezza is likely to be much closer to the current price of injected insulin, albeit there might be a slight premium, similar to that seen with the current pen formulations of insulin.The other major concern that investors have with Afrezza from a commercial perspective is the current lack of a commercial partner. Given MNKD’s current stage as a company, it has no real commercial capability to launch Afrezza on its own, and thus it will either A) have to build it from scratch (a lot of execution risk given the capital, time and expertise required) or B) more likely (and preferred from my perspective), it will partner with a larger pharmaceutical company, one that most optimally is one of the big global diabetes players, such as a Novo Nordisk (NVO), Eli Lilly (LLY) or a Sanofi-Aventis (SNY). For sure there are other companies as well beyond what I would call the «Big Three». These include: AstraZeneca (AZN), which just acquired the part of Bristol-Myers-Squibb’s (BMY) diabetes franchise that it didn’t already co-own through a JV for a total consideration of $5 billion; Merck (MRK), which markets Januvia and Janumet, a combined $6 billion franchise; and Johnson and Johnson (JNJ), which has a large presence in diabetes with its very substantial glucose monitoring business and also its diabetes drug Invokana, an SGLT2 inhibitor that is part of a newer class of agents.But at the moment, Afrezza remains an unpartnered asset. Clearly, at this stage, investors and I would have preferred to have seen MNKD have a global partnership in place by now; however, given how close we are to the PDUFA, my sense is that potential partners are saying that they simply would prefer to see what happens with potential FDA approval. That said, given the totality of the global opportunity there exists for a novel product like Afrezza – and given a rapidly growing diabetes market where the current number of diabetics globally is expected to grow from the current 347 million to potentially as much as 550 million by 2030, we think the likelihood of a commercial partnership for Afrezza remains very high. My guess is that should Afrezza get approved, a partnership of some sort will materialize shortly thereafter.Yoffe: Could you tell us more about the size of the rapid-acting insulin market, and what kind of revenue contribution are you expecting?Dr. Suvannavejh: Overall, we estimate that the current global insulin market is about a $15B market, one that is composed of rapid-, short- and longer-acting formulations of insulin. Within the rapid-acting market, we believe the current market is worth approximately $6B. Based on the work we’ve done, we believe Afrezza can achieve peak sales in excess of $3B worldwide. It’s important to note two things. First, our peak revenue forecast assumes US and ex-US regulatory approvals in both Type I and Type II patients. But given that 90-95% of diabetics are Type II in nature, the vast majority of use and revenue we see for Afrezza would come from the Type II diabetic population. And second, we believe that Afrezza is a type of product that is additive to the current diabetes treatment paradigm, and thus, it could help grow the overall insulin market.Yoffe: In case Afrezza will fail to win FDA approval, what is the value of the rest of the company’s pipeline?Dr. Suvannavejh: MNKD has worked on developing other candidates in the past, including others focused on diabetes and those targeting cancer. However, in order to conserve resources, MNKD has de-prioritized these in the past year or so, even out-licensing two of its three cancer programs. As such, and given very limited visibility, we don’t see any other obvious and tangible pipeline drug candidates. What remains left is a recently announced next generation inhaler device called Sprout that MNKD mentioned on its recent 4Q call. It could be used in acute pain settings, but the reality is that we’ve seen no data for Sprout to date. Therefore, for all intents and purposes, should MNKD be unsuccessful in getting Afrezza approved in the US – which would then probably mean a no-go in ex-US markets – we see little, if not zero, value in the pipeline.Yoffe: How far can MannKind go with the current funding?Dr. Suvannavejh: As many familiar with the MNKD story know, funding has always been big question mark for the company, especially in light of the high expenses that come along with sponsoring large Phase III trials in diabetes. In fact, as MNKD mentioned on its 4Q call last week, its cash burn for the year ending 2013 was a whopping $164M. That said, MNKD has been savvy enough in taking advantage of some unique financing vehicles, including an «at the market» (ATM) sales agreement and a $160M debt financing with Deerfield, a well-known and well-respected healthcare focused institutional investor, that has given it additional runway. And so, as of December 31, 2013, MNKD reported having $71M in cash on its balance sheet, which should be good enough to provide funding into the third quarter of this year. In addition, on March 3, 2014, MNKD announced through its 10-K SEC filing, that it was renewing its ATM agreements with MLV & Co. and another bank for yet another $50 million. That said, we’d hope to see additional non-dilutive cash coming into the company in the form of an upfront payment for any potential licensing deal for Afrezza.Yoffe: As you mentioned, Mannkind renewed its ATM agreement with MLV this month. Do you believe the company will have to use this option to get through its upcoming targets?Dr. Suvannavejh: It’s not a given that they will need to use the ATM, especially if they can sign a big partnership deal for Afrezza. But what’s great about the ATM, in my view, is the flexibility it offers companies. As a reminder, the ATM is a financing vehicle whereby companies are able to issue new common shares into the market, and importantly, at prevailing market prices. Like any traditional follow-on offering, yes, it is dilutive to existing shareholders. But another key benefit of the ATM that it also allows companies to raise money without the typical discounts to the current share price that you often see with traditional follow-ons. Obviously this is a very attractive feature.Yoffe: What is your impression of the management team as leaders?Dr. Suvannavejh: As visionary founder, CEO, and for all practical purposes, the main financial lifeline for MNKD over the past years, Al Mann has been a clear leader of the company. That said, given his age, it’s likely that no matter what the final outcome is with Afrezza regarding potential US and ex-US approvals, new leadership will be needed to steer the company over the next 5-10 years. In my opinion, current President and COO Hakan Edstrom and CFO Matt Pfeffer have both done very solid and credible jobs, and are very likeable. Were I to pick a successor to Al Mann between the two – assuming such a candidate came from internal ranks – I’d probably place my bet with Mr. Edstrom.Yoffe: In closing, what is your rating and price target for MNKD?Dr. Suvannavejh: I’ve had a Buy rating on MNKD shares since I initiated coverage on the company last May. My current price target is $9, which is based on an Afrezza-driven DCF analysis taken out to 2025, assuming a 12% discount rate and 5% terminal growth. My current price target implies there’s additional room for the stock to run.
Parece que hay bastante optimismo esperando la decisión de la FDA…..piano piano…
Ahora mismo esta subiendo bastante …… Habrá alguna noticia?
MNKD
Pequeña subida, pero subida que no hay que despreciar. Esperemos buenas noticias.
Saludos desde el Sur.
PD: ¿Y el SUPER? Dia que no escribe se le echa de menos.
Solo escribo para aportar mi granito de felicitaciones y ánimo al autor por este estupendo Blog, el cual me parece impresionante y que espero, si no mejora, que se mantenga igual de bien.
Llevo muy poquito en el Nasdaq y estoy aprendiendo todo lo que puedo, sobre todo gracias a las entradas y comentarios.
Lo que más me cuesta (podéis llamarme burro…) es interpretar bien los movimientos del pre-market, los cuales puedo ver y operar con mi actual plataforma. Me ponen de los nervios y me descolocan totalmente, pues pienso que no reflejan la evolución posterior del mercado.
Insisto: Felicidades y muchas gracias por compartir vuestros conocimientos.
Ya veremos si tenemos suerte con Mannkind…
ttp://investorshub.advfn.com/boards/read_msg.aspx?message_id=98959326
Estudio de un forero indicando los puntos fuertes de mannkind y afrezza ante el examen de la Fda.
MNKD
Hoy toca caida.
MNKD.-Actualizo su gráfico a este instante en la entrada……….
10% arriba en la preeeee!!!!
Hay news…
AFREZZA Ad comm meeting April 108:59 AM ET • MNKDThe FDA’s Endocrinologic and Metabolic Drugs Advisory Committee meets April 1 to discuss Mannkind’s (MNKD) AFREZZA NDA.The company originally submitted the New Drug Application for its ultra fast-acting inhalable insulin in March 2009. A couple of CRLs and clinical trials later, CEO Al Mann hopes this is the long-awaited breakthrough in light of his $1B+ investment to date.Considering the inherent risk in a binary event such as an Ad comm vote, longs might want to hedge their positions.Shares are up 12% premarket on heavy volume.The firm’s market cap is ~$2B.
MNKD
Ay ay ay que susto, otra vez vuelve a subir.
Dentro con 500 a 5.20…
MNKD
…..y vuelta a caer.
vaya lotería. entrar o no entrar.
MNKD
Ha perforado la pivot alcista que le servia de soporte ¡ay! que nos vamos al infierno.
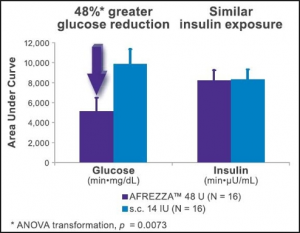
Se habla de lotería para aprobar en el tipo I de diabetes y hay más optimismo para que se apruebe para el tipo II. Yo desde luego ya no abandono el barco. Que sea lo que Dios quiera, pero una insulina inhalada, a poco que iguale los resultados de una inyección, debería ser valorada muy positivamente. Desde luego y por lo que estoy viendo, para tocar el cielo hay que conocer muy de cerca el infierno. Por el bien de la medicina y sobre todo de los diabeticos, esperemos que haya suficiente potencial para aprobar afrezza, y que las grandes pharmas jueguen limpio y no quieran hundirlo con sus $$$$$..Ya veremos…
yo creo que la aprobaran pero como pasó en chtp una buena barrida meterá todavía.
Ayer viernes cerré la posición corta que tenía abierta. No me la juego. Yo creo que la van a dar el si, pero me abstengo de entrar hasta que no termine esta semana.
http://translate.googleusercontent.com/translate_c?depth=1&nv=1&rurl=translate.google.co.jp&sl=auto&tl=es&u=http://insulinnation.com/treatment2/artificial-pancreas/inhalable-insulin-a-breathtaking-development/&usg=ALkJrhi73Nt1mCyNFpFJWropQaLzVEOozQ
Lectura muy interesante para el que quiera conocer un poco mas la Historia de Mannkind y afrezza. El avance que supondría la introducción de este fármaco en nuestras vidas seria incalculable, ya no solo para tratar la diabetes, si no que podría abrir las puertas al tratamiento de otras enfermedades. GO AFREZZA!!
http://translate.googleusercontent.com/translate_c?depth=1&nv=1&rurl=translate.google.co.jp&sl=auto&tl=es&u=http://www.beyondproxy.com/mannkind-corporation/&usg=ALkJrhjUN11CFgCvWMzUjfnOLI-8G6orkA
Y otra mas, muy completa, que ya firmaba yo que se cumpliese. Pero esto es un mundo de intereses y pirañas, que hace que cada palabra haya que cogerla con pinzas…
Hola !
Llevo 2000 papelinas compradas en el verano del 2012.
Ha llegado a estar la acción 200% por encima del precio comprado pero si crees en algo, hay que dejarlo correr unos años, es la forma de multiplicar de verdad.
Acertaremos con Mannkind ? Solo el tiempo lo dirá.
Para bien o para….mejor ? hemos llegado hasta aquí y esperamos.
Suerte -que también es necesaria- el 14-04-14…bonita cifra, verdad ?
José Manuel
hoy quien se reunion el panel de expertos? hoy es clave porque si sale negativo el 14 no habrá nada y mañana mismo habrán meetido el palo. si hoy sale positivo mañana habrá una buena revalorización.
claro que hoy habrá como siempre manipulación y seguramente la tiren por rumores que si no la aprobarán que si tal y cual. lo de siempre
La reunión es mañana, todo lo que pase hoy
Pecata minuta
…..
.
Bueno….cerrado el gap alcista del nueve de mayo pasado, ahora…..esperemos que doble en breve.
Si fuera antes del 14, mejor que mejor je, je
MNKD
http://www.thestreet.com/story/12561947/1/mannkind-the-best-afrezza-fda-panel-preview-ever.html
Sacado del mismo artículo,,,,
Then, FDA raised concerns about Afrezza’s lung safety, including a small, but higher incidence of lung cancer seen in Afrezza patients.
Should people be freaked out about the FDA raising concerns about a lung cancer risk in Afrezza patients?
FDA found a numerical imbalance in reported lung cancers going against Afrezza, but the incidence rate is small and animal studies showed no cancer signal. Insulin is a growth factor and a similar lung cancer concern was raised about Pfizer’s (PFE_) discontinued inhaled insulin Exubera, so I can understand why this will be a significant topic on Tuesday. However, it doesn’t seem like a knock-out punch against Afrezza.
Muy curioso,,,desconozco como se planificaron y ejecutaron los estudios pero tampoco explica que en el grupo de Afrezza podía haber mas personas con factores de riesgo para desarrollar cáncer de pulmón que en el otro grupo,,,
MNKD
¡Vaya tortazo! ¿por que?
MNKD.-Les dejo su gráfico actualizado en la entrada y les deseo suerte en el envite, porque la manipulación es tremenda, ¡como siempre!.
MNKD
¡Ay! con 11000 papelillos y esto despeñandose. Tecnica WB: Uuuuummmmm -vestido de tunica azafran y rapado-.
Gracias SUPER por tus desvelos.
Toda la suerte del mundo Josej
tengo muchas dudas que aprueben afrezza,no veo estudios clinicos concluyentes aunque esto es una loteria
en el caso de drtx si que era muy evidente la aprobacion
No actualiza el pre mark? Lo veo igual desde las 12:00 a 4, 2 $ y 675 cromos….. es raro no?
esta suspendida ??
TAU : tu que estás muy puesto siempre, sabes lo que sucede hoy ?
Supongo estará suspendida, pero….por qué ?
Gracias
MNKD
Hoy es la reunion del panel de expertos previo a la aprobacion por la FDA.
digo yo que si no lo aprobaron dos veces, a la tercera ya irán con los deberes echos. tendrían que ser muy tontos si saben que las van a rechazar presentarse sabiendo que les tumbarían la cotización. no creeis?
pues como la tengan suspendida de cotización pinta muy pero que muy mal. Siempre que me han pillado con una suspensión en la cotización me he llevado un buen tortazo.
Espero que no sea así.
MNKD
Parece que el panel votó a favor de la tipo I (13-1-1) 1 en contra 1 abstención,,,,
MNKD
Parece que el panel votó a favor de la tipo I (13-1-1) 1 en contra 1 abstención,,,,
Parece que el panel votó a favor de la tipo I (13-1-1) 13 si, 1 no ,1 abstención,,,
MNKD
Tipo 2 14-0
Me alegro por todos los que estáis pues espero que el subidón sea mínino y por el que escribió el artículo el día 31 que debería estar investigado,,,,